
Critical questions
During COVID-19, Every Breath Counts Coalition members met weekly to mobilize support to the countries struggling to meet the surging demand for medical oxygen, pulse oximeters, and related medical devices and therapies.
During those conversations, tough questions were asked and debated. We captured the responses to the most ‘critical questions’ that emerged during that intense period so we could all learn lessons and prepare better for the next respiratory pandemic.
Below you will find a collection of the questions and answers that remain relevant as we mobilize the medical oxygen supplies needed to save lives now and prepare for the next respiratory pandemic, which experts predict is likely in the next decade.
By learning the lessons from COVID-19 we can prevent a repeat of the tragic 2020 and 2021 oxygen shortages that elevated mortality and caused so much suffering to patients and families all across the world.
How to protect maternal, newborn, child and adolescent health and nutrition services in low- and middle-income countries during the COVID-19 pandemic?
Answer provided by John Borrazzo, Senior Health Specialist at the Global Financing Facility (GFF), which is hosted by the World Bank.
As the COVID-19 pandemic escalates in low- and middle-income countries, there is a growing risk of widespread disruptions in access to maternal, newborn, child and adolescent health and nutrition services.
A pulse survey of the 36 GFF-supported countries found that nearly half are already reporting life-threatening service disruptions. The findings revealed that the COVID-19 pandemic is disrupting delivery of essential services such as ante-natal care visits, attended births and delivery of child vaccinations. These disruptions are likely to lead to significant increases in preventable maternal and child illness and death.
There is also the beginning of systematic problems such as the ability of families to secure transport to health facilities. Limited supplies of personal protective equipment for health workers result in both risks to the health workforce delivering RMNCAH-N services as well as suppressing demand for facility-delivered services because of fears of virus transmission.
The COVID-19 pandemic is likely to be an ‘inequity multiplier’ – disproportionately affecting availability, accessibility, and utilization of RMNCAH-N services by the poorest and most vulnerable populations.
It is vital that countries do not fall backwards on the improvements that have been made to maternal, newborn, child and adolescent health and nutrition services in recent years.
The GFF is protecting essential health and nutrition services in GFF partner countries as part of the COVID-19 response by:
- Supporting frontline healthworkers, through flexible funding and technical assistance, for training and other activities that complement efforts by the World Bank Group and other partners to supply personal protective equipment (PPE);
- Supporting leaders in eligible countries to develop cross-cutting plans to maintain essential services during the pandemic.
- Supporting effective coordination of financing – such as through resource mapping and expenditure tracking to ensure resources are used efficiently.
- Sharing good practices, lessons learnt and guidance among leaders and health workers, including the latest information to inform service delivery, using existing GFF knowledge and learning support to Country Platforms.
- Providing technical support to leverage COVID-focused investments to ensure availability of critical supplies that also support RMNCAH-N, such as sustainable oxygen systems and blood supply.
- Assisting countries to introduce service delivery changes – such as by separating patients seeking routine health care from those with COVID-19, scaling up the use of telemedicine and other digital health channels.
- Developing strategies to ensure that essential health products reach local communities, including via partnerships with the private sector.
- Developing innovative approaches to social and behaviour change communication to help communities respond to the pandemic and continue to access critical RMNCAH-N services.
- Supporting actions to avoid disruptions to supply chains for essential RMCNAH commodities.
To find out more read: “Emerging secondary global health crisis from disruptions in primary health care due to COVID-19” (23 April 2020).
How do we prioritize invasive versus less-invasive respiratory therapies in low-resource settings?
The answer is provided by Tim Baker, a critical care doctor affiliated with the Karolinska Institutet in Sweden and the Muhimbili University of Health & Allied Sciences in Tanzania.
The headline figures of intensive care unit (ICU) requirements for COVID-19 patients in wealthy countries are masking the need for essential care in low-resource settings.
Expensive, high-tech equipment demands highly trained health workers and an extensive supply of medical oxygen and, moreover, serves a tiny proportion of the population. This is not feasible in low-resource settings and can be dangerous if not properly supported.
Indeed, most critically ill patients in poorer countries are cared for in emergency units and general wards and do not have access to advanced care in ICUs. Data from hospital wards in Malawi showed an alarming 89% of hypoxic patients were not receiving oxygen. Such capacity cannot support widespread mechanical ventilation.
We need to move away from high-tech mechanical ventilation to low-cost essential care which serves a larger proportion of the population. This requires a comprehensive integrated approach – where oxygen is the key element that helps many diseases – and support to ongoing health services provisions to non-COVID patients.
Essential care in low resource settings should include the following elements:
- Improving community case management and hospital readiness;
- Ensuring good quality clinical practice for both identifying and caring for critically ill patients;
- Hospitals establishing effective systems for triage and essential care in emergency units and wards, including patient separation and staff safety;
- Emphasizing the central role of oxygen therapy – and ensuring oxygen supplies and delivery systems and guidelines for sustainable and appropriate use;
- Head-up or prone patient position, suction, and simple chest physiotherapy; and
- When human resources are limited, ensuring less trained health workers or vital signs assistants implement and share such care.
Quality essential care of critical illness could have a significant impact on reducing COVID-19-related mortality even without ICUs. We must urgently enhance the ability of health services in low-resource settings to provide good quality essential care of critical illness.
For further information see: Essential care of critical illness must not be forgotten in the COVID-19 pandemic, The Lancet, 1 April 2020
How can international actors best support local manufacturing of WHO-recommended COVID-19 technologies, where appropriate and feasible?
The answer is provided by: Dr Paulin Banguti, University of Rwanda, King Faisal Hospital, and the Rwanda Society of Anesthesiologists.
As the world scrambles to buy ventilators in the COVID-19 pandemic Rwanda has come up with its own solution: the first locally produced ventilator.
After observing how the pandemic has ravaged the world, and the importance of oxygen in the treatment of the virus, biomedical engineers, from the Integrated Polytechnic Regional Centre in Kigali, challenged themselves to produce the ‘Rwanda Emergency Ventilator’.
The ventilators are made from locally sourced materials and can be produced at a much lower cost. They are also more appropriate for low-resource settings – the ventilator does not need oxygen tanks (it uses natural oxygen), can run on a battery and is easily maintained.
The engineers have produced two types of ventilators – for hospitals and portable use in ambulances. The plan is for the ventilator to be made available at an affordable market price, and the prototype is now being shared with other low- and middle-income countries.
The COVID-19 pandemic has underlined the need for cost-effective local solutions.
But there are many challenges. It is difficult to purchase components (such as sensors), as global supply chains become overloaded, and oxygen supply is limited. Support to train technicians, clinicians and health workers to use and maintain the equipment, is hard to find.
Rwanda has the technical expertise – the engineers, scientists, physicians and IT developers – to design and test COVID-19 technologies. What is urgently needed now is support from governments, donors and private investors to manufacture and distribute them at scale. With this support – financial and technical – Rwanda would not only be in a stronger position to control the pandemic, but it would ensure that respiratory care technologies are available in the health system for the long term.
ENDS…/
For further information watch Dr Banguti and the team of biomedical engineers from the Integrated Polytechnic Regional Center (IPRC) in Kigali discuss the first locally-made ventilator
In you have an innovative product that is approved for emergency use or already commercially available, submit your product for consideration to WHO: https://www.who.int/medical_devices/priority/COVID-19_innovations/en/
Contact techinnovation@who.int
What do we really know about access to pulse oximetry and oxygen in low- and middle-income country health facilities?
Answer provided by Hamish Graham, Murdoch Children’s Research Institute (MCRI), and the Centre for International Child Health, University of Melbourne
The bad news: it is worse than we think.
A recent study of 12 Nigerian hospitals found that more than 90 percent of hospitals surveyed said they had oxygen supplies. But closer examination at the ward level revealed that:
- Just five hospitals had oxygen available in paediatric wards, and just one used pulse oximetry for paediatric care.
- Only three of 57 oxygen concentrators that were tested were producing medical‐grade oxygen while 24 were simply blowing out air.
- Less than one in five children with pneumonia received the oxygen they needed. We estimated that this had contributed to 300 excess deaths over the 2-year study period.
It is the same story in other parts of Nigeria and especially in the remotest health facilities of northern Nigeria.
Patients who may benefit from oxygen do not receive it – either because oxygen is not available or because their need for oxygen is unrecognized. And where oxygen is available, it is expensive and out of reach to most people on a Nigerian salary (costing $25 USD per day in hospitals we surveyed). Furthermore, healthcare worker skills and confidence in using pulse oximetry and oxygen is low – especially in health centres and smaller hospitals where most sick children first present.
Access to oxygen is a big problem for children suffering from pneumonia but it is also critically important for children, and adults, with a range of other medical and surgical conditions. In fact, almost one-quarter of neonates and one in 10 children (aged 1 month to 15 years), admitted to these Nigerian hospitals had severely low blood oxygen levels (hypoxaemia, SpO2<90%). And these patients were seven time more likely to die than those without hypoxaemia.
The good news: affordable solutions already exist.
Our oxygen implementation project in Nigeria is one of a number of recent projects showing that low-cost oxygen solutions can be effectively implemented. In our case, we used oxygen concentrators, combined with flowmeter stands and simple plastic tubing to share oxygen to multiple patients simultaneously. We introduced simple guidelines, practical training, and brought technicians, healthcare workers, and hospital managers together to address a shared headache. There were lots of obstacles to overcome, like poor power supply, overworked staff, and hospitals that were already in debt. But the multidisciplinary teams were able to create change and it had a huge impact.
We found that oxygen access for children increased substantially, and we detected a reduction in child pneumonia mortality (though not all-cause child or neonatal mortality). But there were some surprises.
First, we found that simply introducing pulse oximetry as a standard practice (the “fifth vital sign”) dramatically changed oxygen access, even before we introduced the new oxygen equipment. In fact, the proportion of hypoxaemic children receiving oxygen increased from 20% to over 80% within a few months of hospitals making pulse oximetry part of standard care. The right patients were now being identified, and staff were mobilising limited oxygen resources to get it to them. This translated into a 50% reduction in risk of death from pneumonia. Of course, pulse oximetry alone cannot help those hospitals that do not have any oxygen – but it should be a reminder of the critical importance of pulse oximetry to promptly detect hypoxaemic.
The second surprise was that the amount of oxygen used actually went down in some hospitals. Patients were now receiving lower flow-rates (just the amount needed) and oxygen was not being wasted on patients who didn’t need it. This is important, as more efficient use of oxygen can reduce the enormous cost burden on hospitals (and patients).
Key takeaways
- When we talk about oxygen access – we need to be measuring at the level of patients. It is easy to survey hospitals about what gear they have – but this misses the problems with maintenance systems (skills, access to spare parts) and clinical systems (pulse oximetry, protocols, cost) that are currently preventing patients from getting oxygen.
- If we want to improve oxygen access – pulse oximetry is absolutely essential. We should not be funding, supplying, or training on oxygen without pulse oximetry. Oxygen without pulse oximetry is dangerous and wasteful.
- When we think about oxygen costs – we need to think whole of life cycle, and think what is appropriate for what context. Oxygen suppliers must be more transparent about pricing (many hospitals get tied into costly oxygen subscription programs because they are cheap to sign up). Oxygen concentrator and plant manufacturers must be honest about warranties and repair needs (often we cannot get spare parts, or we pay to return to manufacturer).
- Hospitals need to do better with what we already have. Start with implementing pulse oximetry and basic clinical protocols. Support technicians to revive, maintain, and extend the reach of existing oxygen equipment. Then think about smart ways to improve your oxygen systems for the longer term – whole of hospital, clinical and maintenance perspectives, fully funded.
ENDS…/
Oxygen for Life Initiative has updated its open-source oxygen resources and oxygen systems installation guides. The guide includes planning guidance for adult and children’s wards, targeting district-level hospitals.
For more information, read some of the latest research:
- Oxygen therapy for children: A key tool in reducing deaths from pneumonia…(2020)
- Improved oxygen systems at hospitals in three Nigerian states… (2020)
- Oxygen systems to improve clinical care and outcomes for children and neonates…(2019)
- Providing oxygen to children and newborns: multi-faceted assessment…(2019)
- Hypoxaemia in hospitalised children and neonates…(2019)
- Adoption of paediatric and neonatal pulse oximetry by 12 hospitals in Nigeria…(2018)
- Providing oxygen to children in hospitals: a realist review. (2017).
How do we ensure that COVID-19 respiratory therapies are introduced in ways that strengthen the ability of LMIC health systems to reduce deaths during the pandemic and beyond?
Answer provided by Robert Neighbour, Managing Director, Diamedica, a UK-based supplier of anaesthesia, ventilation, ICU and oxygen supply systems to low resource settings.
Ventilators can play a vital role in the management of patients with a severe respiratory illness such as COVID-19 and pneumonia. But the scramble for mechanical ventilators for low- and middle-income (LMIC) countries has distorted what constitutes an effective COVID-19 response for countries with under-funded health systems.
At the same time, the rush by manufacturers in high-income countries to produce ventilators from objects such as vacuum cleaners, windscreen wipers and car parts (many not going beyond the initial prototype) has resulted in ventilators of low efficacy being produced and finding their way to LMICs.
Providing an unsupported and uncontrolled medical device, possibly manufactured by those with insufficient understanding of the implications, is dangerous. We need to get the right equipment to LMICs.
Ventilators should only go to countries and locations where health systems are able to absorb them. Many countries do not have the required infrastructure to run such complex machines; oxygen provision is patchy or unavailable; clinical guidance, training and support is limited; and health workers are not trained to use many of the advanced technologies. Moreover, experience in the United States and Europe has shown that health outcomes from mechanical ventilation are often poor for COVID-19 patients.
It is crucial that we are aware of local conditions so that we are not providing equipment that is unsuitable from the start. Oxygen concentrators intended for use in countries with limited resources should be evaluated before they are purchased; no oxygen concentrator should be supplied without voltage protection to locations where electricity is unreliable as both power outages and surges can damage machines, leaving patients without oxygen. Similarly, non-invasive therapies such as Continuous Positive Airway Pressure (CPAP) and Bi-level Positive Airway Pressure (BiPAP) require large oxygen flows for adults and may not be suitable for many low-resource settings that do not have reliable and affordable access to large supplies of medical oxygen.
I believe that basic oxygen therapy will provide the best outcomes for the majority of patients, with lower risks to clinical staff. This can be provided from oxygen concentrators using high-flow nasal cannulae or oxygen masks with monitoring by pulse oximetry. This, together with personal protective equipment, will have the biggest impact on reducing mortality from COVID-19 in low-resource settings.
Fundamentally, we need to focus on the best health outcomes for the largest proportion of the population and we must not forget the existing unmet medical needs of LMICs whilst focusing on equipment for the pandemic.
ENDS…/
Read about Diamedica’s COVID-19 response here.
Article by Robert et al Evaluation of Oxygen Concentrators for Use in Countries With Limited Resources: https://pubmed.ncbi.nlm.nih.gov/23654218/
Oxygen with Limited Resources: Oxygen Therapy with Limited Resources and Wall Chart
How can we ensure oxygen concentrators supplied as part of the COVID-19 response become lasting assets for health systems to improve access to oxygen therapy?
Ebrima Nyassi, biomedical engineer from The Gambia, shares his tips.
Oxygen concentrators have become an important component of the COVID-19 response. With flow rates of up to 10 litres per minute they can be used to treat patients with severe illness who do not need ventilation. They are easy to install and provide an instant source of oxygen – where electricity is available. But in low-resource settings, oxygen concentrators are often found to be in disrepair – either blowing out air rather than concentrated oxygen or lying around unused for lack of spare parts.
How can we make oxygen concentrators more sustainable?
As a Biomedical Engineering Technologist (BMET) at the Medical Research Council (MRC) I participated in the establishment of the Health Technology Management Program of skilled BMETs. We carried out inventories and managed equipment at MRC’s 42-bed hospital and partner government health facilities. We conducted a retrospective analysis of the preventive maintenance and repair records for 27 concentrators over a seven-year period to determine the common causes of failure; replacement parts; equipment downtime; expected useful lifespan; and annual repair costs.
Two encouraging findings emerged from our work:
Firstly, we showed that oxygen concentrators can operate efficiently for six years – or more.
- Over a seven-year period, 85% of concentrators experienced just two or fewer repairs; the average time before the first failure was more than two years. The median age of the concentrators was 6.1 years.
- The technicians successfully resolved 94.6% of the 56 reported faults, with around 70% requiring less than one day of downtime. The majority of concentrator faults (filter and valve replacements) were repairable for less than $US10 on average.
Secondly, the skills needed to repair the most common failures are quite basic and could easily be taught.
- Through a skill-mapping analysis we identified 31 basic technician skills that would be sufficient for the repair of more than 90% of observed oxygen concentrator failures and for routine preventive maintenance. Most of these skills are drawn from the library of Biomedical Technician Assistant skills.
How can we replicate this model in other low- and middle-income countries (LMICs)?
We have built a model for health technology management in LMICs, where trained biomedical technicians are equipped with the required skills and tools to maintain essential oxygen equipment for COVID-19 and beyond.
Short-term steps for concentrators deployed for the COVID-19 response include:
- Log and track all devices and maintenance activities in an inventory system. This can be just at the facility level and with basic Excel to record and track inventory and maintenance history. You do not need complex and expensive software.
- Empower clinical staff with the knowledge and training for basic user care and to notify technicians when they spot signs of malfunction. It is recommended that hospital management/administration formally make this part of the role of clinical staff.
- Dedicate a lead engineer/technician to take charge of the services with the support of colleagues and management.
- Establish a six-monthly preventive maintenance schedule (three months in dusty areas) to assess the quality of oxygen being produced with an oxygen analyzer and timely maintenance.
- Ensure availability of spare parts/consumables (e.g., air Intake filters, bacteria filters, extra nozzles, sieve beds, humidity bottles, main PCB board, solenoid valves etc.).
- Ensure availability and easy access of service and user manuals.
Long-term steps – ensuring sustainability beyond COVID-19 include:
- Maintenance starts from the point of purchase!
- Engage in-house engineers in planning and procurement decisions to ensure procurement proposals for concentrators include the following:
-
-
- Warranty
- Spare parts
- Appropriate tools and test equipment such as oxygen analyzers (about $US700)
- Service manuals
- Any training gaps needed
-
- Select quality concentrators that can work in hot, humid and dusty conditions
- Advocate for building a workforce of biomedical engineers and technicians within the health system, and help get them the recognition they deserve as essential healthcare workers.
ENDS…/
Additional resources:
- Engineering World Health Biomedical Equipment Technician (BMET) Library
- Assist International HTM webinars on Oxygen Concentrators: Part 1 and Part 2
How can we support low- and middle-income country governments to equitably deliver COVID-19 vaccines?
Read answer from Trevor Zimmer, COVID-19 Vaccine Equity Project
While 104 million doses of COVID-19 vaccine have been administered worldwide, just 25 doses have been given in one low-income country. The WHO Director-General, Dr Tedros, warned that this massive disparity placed the world on the brink of a “catastrophic moral failure”, as rich nations buy up vaccine stocks leaving low- and middle-income countries (LMICs) struggling to find supplies.
Although several LMICs are now procuring vaccines directly from manufacturers, including from the Serum Institute of India, the rollout of vaccines to LMICs through the Access to COVID-19 Tools Accelerator (ACT-A) COVAX Facility will begin in late February. The goal of COVAX is to distribute 2.3 billion doses of COVID-19 vaccines in 2021 enabling lower income countries to protect 27% of their populations.
We also need to address vaccine equity within countries
The COVID-19 Vaccine Equity Project (CVEP) was set up to address COVID-19 vaccine equity within countries. The project supports LMIC governments and frontline immunization workers to equitably prepare for COVID-19 vaccine programmes – to ensure the vaccines reach the most vulnerable populations to prevent the most deaths.
Specifically, the group of technical experts assess ‘preparedness’ (including resource allocation, supply chains, target populations) and co-create or inform country-led action plans that reflect the realities of local service delivery. The project works closely with relevant government ministries in coordination with ACT-A, Gavi Country Support Teams and other actors – including the Sabin Vaccine Institute’s Boost Community to reach frontline immunization workers.
The project seeks to support up to 16 countries address their equity needs related to vaccine access, with a focus on countries participating in the COVAX Facility Advanced Market Commitment (AMC). As a first step, the project has engaged a subset of countries – Ecuador, Ethiopia, Kenya and Nepal – in a pilot. Each country represents different needs (e.g., prioritising vulnerable populations in Ecuador) and different funding modalities (three countries are funded through the AMC; Ecuador is self-financing).
In addition, CVEP – in collaboration with COVAX country readiness and delivery (CRD), Project ECHO, TechNet and the Boost Community – will be launching a community of practice (CoP) to provide immunization professionals opportunities to engage in informal, facilitated discussions on themes related to equitable access. The CoP will also be a channel to share ‘global goods’ emerging from CVEP’s country-specific support, and to provide insights from the frontlines to global and regional actors engaged in vaccine introduction.
The aim of the pilot is to generate best practice guidance and to share peer-to-peer learning. Following the successful pilot, the partners have secured additional funding to scale the project to 12 more countries 2021; with the goal of reaching 45 countries.
Crucially, the project is focused on preparedness to ensure that LMICs are ready to roll the vaccines out effectively. The goal is to amplify partner efforts and ensure technical assistance is tailored to country-level priorities and needs to ensure that the vaccines truly reach the poorest and most vulnerable populations.
ENDS…/
The COVID-19 Vaccine Equity Project is a joint initiative of the Sabin Vaccine Institute, Dalberg and the JSI Research and Training Institute – with support from the Skoll Foundation. It works in close collaboration with the Access to COVID-19 Tools (ACT) Accelerator.
Our World in Data tracks the current state of COVID-19 vaccine coverage in LMICs here.
How many COVID-19 deaths in LMICS are the result of lack of access to medical oxygen?
Read answer from Dr Hamish Graham, Murdoch Children’s Research Institute (MCRI) and Centre for International Child Health, University of Melbourne
Oxygen and pulse oximeters are critical components in providing quality care for patients with COVID-19 and other respiratory illnesses like pneumonia, and is an essential medicine for safe surgery, anaesthesia and obstetric care. So why are current methods of measuring oxygen access still inadequate?
Current methods of measuring oxygen access massively underestimate the problem
The COVID-19 pandemic has exposed global oxygen system deficiencies and major gaps in how we understand and measure ‘oxygen access’. We still do not have national estimates of medical oxygen access or the tragic consequences of lack of it. We don’t even know the number of hypoxic patients, let alone what percentage actually receive oxygen.
In our new study “Measuring oxygen access: lessons from health facility assessments in Lagos, Nigeria” we investigated oxygen access in 58 health facilities in Lagos state, Nigeria.
Of 58 hospitals and primary health care centres surveyed, we found huge gaps and differences in access across facilities including:
- Just 8 facilities had a functional pulse oximeter.
- Just over half (59%) facilities had a functional source of oxygen available, of which most (91%) facilities had it available in a single ward area, typically the operating theatre or maternity ward.
- Oxygen services were free to patients at primary health centres but expensive in hospitals and private facilities.
- No facilities were equipped to meet minimum oxygen demands (based on existing data).
Our study argued that improving access to oxygen will require major changes in how access is measured. Health facilities need to ask themselves not only is oxygen available, but does it work, is it reaching patients, and is it affordable? If they answer no to any one of these questions, oxygen access is compromised.
In short, access to medical oxygen is a function of three factors: 1) use; 2) availability; and 3) cost.
1) Use. Oxygen therapy should be guided by pulse oximetry and clinical guidelines
Ideally, we would like to know what proportion of patients who need oxygen actually receive it. The next best indicator for oxygen reaching patients is pulse oximetry coverage. Pulse oximetry is essential for triaging unwell patients and guiding oxygen therapy and should be used for all acutely unwell patients coming to hospital. If pulse oximetry is not performed routinely then health managers can be sure that oxygen is not reaching the patients who need it and existing oxygen supplies are being wasted
2) Availability. Functional pulse oximeters and oxygen supplies at point-of-care
It is not enough to rely on the presence of an oxygen source (despite this being the standard metric) as it does not necessarily correlate with actual access to patients. Oxygen should be available in all parts of the health facility where hypoxic patients are treated (not just where anaesthesia is performed) and minimum functional requirements for oxygen sources (e.g., purity) must be assessed by at point-of-care, as oxygen concentrators, cylinders and pulse oximeters may be present without being in working order.
3) Cost. Cost to patients and cost to hospitals
The cost of providing oxygen – to hospitals and patients – must be documented as there are examples of functional oxygen being available in hospitals but tragically unaffordable to patients or burdening facilities with increasing debt.
We conclude that a multifaceted approach to measuring oxygen access that assesses access at the point-of-care and ideally at the patient-level is critical.
We also propose standard metrics to measure and report oxygen access – across the three domains of use, availability and cost – and describe how these can be integrated into routine health information systems and existing health facility assessment tools.
Our proposed indicators include: (1) % of acutely unwell patients being screened with pulse oximetry, (2) % with hypoxemia receiving oxygen therapy, (3) % of wards with functional pulse oximetry (4) % of wards with functional oxygen delivery, (5) daily oxygen cost to patients, and (6) annual hospital expenditure on oxygen equipment, maintenance, and repairs.
Routine measurement of these three domains of oxygen access will accelerate local, regional and global efforts to guarantee life-saving oxygen to all those who need it during the pandemic and beyond.
ENDS…/
For more information see: “Measuring oxygen access: lessons from health facility assessments in Lagos, Nigeria,” 3 August 2021, Hamish R Graham, Adegoke Falade, Ayobami A Bakare, Omotayo Olojede, Oyaniyi Olatunde, Obiomo C Uchendu et al.
Every Breath Counts has also called for a global effort to count COVID-19 deaths due to lack of medical oxygen. The coalition also encourages health facilities to adopt the access to oxygen metrics described above.
How can we openly, transparently and independently measure the accuracy of pulse oximeters, including on different skin tones?
Read answer from Dr Michael Lipnick, University of California San Francisco
Pulse oximeters are essential healthcare tools that measure oxygen saturation in the blood. They are a strong predictor of severe illness and pneumonia death – including from COVID-19 – and are therefore a gateway to safe and effective oxygen use.
But despite their lifesaving potential, these simple tools are not available in many health facilities in low-resource settings. And where these devices are available, they may not perform safely – some pulse oximeters are more accurate and robust than others, and some may overestimate oxygen levels in darker skin tones, with potentially tragic consequences.
The COVID-19 pandemic has highlighted these inequities. There has been a surge in the number of low-cost oximeters in low- and middle-income countries, which has certainly increased access, but which may not have improved treatment outcomes.
Inaccuracy of pulse oximetry in patients with darker skin tones has been long known but is now garnering attention. Yet, the extent of these inaccuracies, the impact on patient care, and the reasons for such inaccuracies are not fully understood and demand urgent attention.
All pulse oximeter manufacturers are required to conduct laboratory-based studies of oximeter performance in patients with varying skin tones in order to receive FDA/ISO certification. However, many oximeters in clinical use do not undergo such testing, and for those that do, there are major limitations in current testing protocols that allow some oximeters to perform well in the lab but not in the clinical setting.
Patients, their families, and health workers must have confidence that pulse oximeters will be both available and reliable when they measure oxygen saturation, regardless of skin tone and other differences in patients and their surroundings.
The OpenOximetry.org Project
The OpenOximetry.org Project is a collaborative effort to build an open-access repository of independent assessments of pulse oximeters being led by the Hypoxia Lab (which has published more than 400 peer reviewed manuscripts) and the Center for Health Equity in Surgery and Anesthesia, at the University of California San Francisco.
Currently, the website allows you to compare pulse oximeters on a range of measures – including by manufacturer, by oximeter type, by certification, by features, by patient population and by performance, based on laboratory studies. With greater investments in expanded laboratory protocols and real-world clinical trials these performance assessments could be further improved to help guide donors, users and manufacturers seeking safe oximeters for patients care.
Our assessments have revealed poor performance in a large number of pulse oximeters, particularly finger-tip pulse oximeters that are available for the lowest prices. Current regulations and standards for manufacturers are inadequate and need to be strengthened to improve the quality of pulse oximeters.
Setting a higher bar for performance
In our commentary, Pulse Oximeter Performance, Racial Inequity, and the Work Ahead, we call on leading regulatory agencies to modify standards for (1) measuring accuracy and precision of oximeter performance; (2) analyzing and reporting performance outliers; (3) diversifying study subject pools; (4) thoughtfully defining skin pigmentation; (5) reporting data transparently; and (6) accounting for performance during low perfusion states.
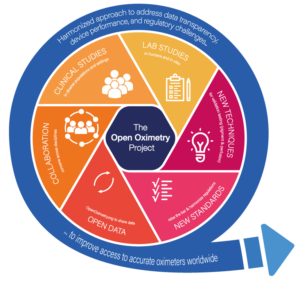
In addition to openly sharing data on independently tested oximeters, The OpenOximetry Project will create new lab techniques to better predict real world clinical performance, establish better clinical studies protocols to improve interpretability of real-world clinical studies, and utilize data to inform new regulatory certification processes.
A multipronged approach will help reduce bias in pulse oximeter performance and improve access to safe oximeters across the globe.
ENDS…/
*****
For more information see:
The OpenOximetry.org Project here
UCSF Hypoxia Lab publications here
Pulse Oximetry Performance, Racial Inequity and the Work Ahead here
Racial Bias in Pulse Oximetry Measurement here

